Michael Joe Cini
19th April 2021
Hensler Surgical Technologies’ Bone Press Secures CE Mark
Hensler Surgical Technologies’ bone press receives CE mark for its Hensler Bone Press (HBP) used for bone fusion surgery
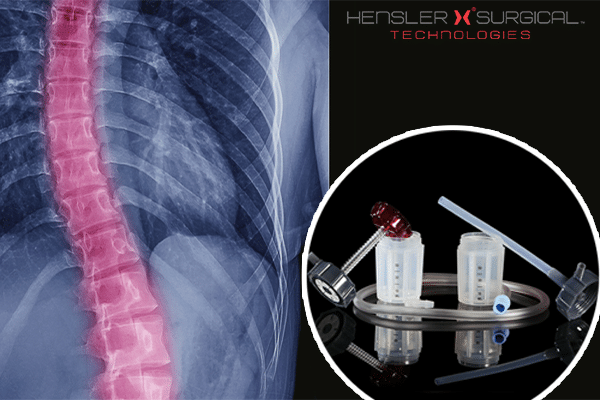
Hensler Surgical Technologies has secured CE mark for its Hensler Bone Press (HBP) – used in bone fusion surgery. The HBP can harvest high speed drilled autologous material during bony decompressions, and orthopaedic surgical procedures.
The company has also noted that the device can be used in a host of other procedures including but not limited to: vertebral partial/full corpectomies, scoliotic deformity, in-situ-fusions, orthopaedic non-unions and foot and ankle surgeries.
The HBP is noted for being disposable and user friendly, and essential for surgeons who intend on using a patients’ own autologous material for fusion cases.
Launched in 2012, the HBP is currently being used for surgery in the U.S as well as respective foreign countries who don’t comply with the CE mark.
Hensler Surgical CEO and inventor Sean Hensler said in a statement:
Harvesting drilled graft material is an arduous but necessary undertaking to improve fusion outcomes in a wide array of surgical procedures.
We are proud to offer a device that procures this graft material quickly, allowing for the procurement and processing of this critical and valuable autologous resource into a moldable and malleable graft for use in fusion surgeries.
Our universal device excels at high yield graft recovery designed to flow seamlessly in the operating room setting for all staff that uses it.
Source: Medical Device Network




